Please select your Region.
Please select your Region.
Sep 17, 2014
ARKRAY, Inc. (herein 'ARKRAY') has established a new medical device/reagent brand for animals "thinka" and is due to release the products for animals starting from 27 September.
The brand name "thinka" means to "think animals" and represents our desire to "think" about the health of animals and "evolve" (shinka in Japanese) the animal medical care. We will continue to work toward commercialization of thinka products using our technology we have built up over many years in the developement of human medical devices/reagents for diabetes and urine testing.
ARKRAY will release the "thinka Canine Heartworm Ag Test Kit CHW" (canine heartworm) on 27 September as the first thinka product. The thinka Canine Heartworm Ag Test Kit CHW is to test antigen of filarial (heartworm) infection*1. Only application of 2 drops of blood sample is required for this one-step testing kit. It allows easy and quick filariasis antigen test during the busy season.
The animal medical and diagnostic market is currently reaching 300 billion yen*2 and increasing every year. In line with the increase of the number of pets, the number of animal hospitals is on the rise to about 11,000 facilities*3. ARKRAY continues to use its core technologies to develop new products and services in the animal medical care field and strengthen its business areas.
*1 Filariasis is a parasitic disease that is caused by thread-like worms that occupy the heart and lung artery of a dog. They are spread by blood-feeding mosquitoes. The preventive agent for the disease is dispensable on directions or prescriptions after an examination and blood testing by a veterinarian.
*2 Source: "Introduction of the animal hospital market" published by JPR Corporation on 27 February 2009
*3 Source: "Establishment status of treatment facilities for pets 2013 (No. of facilities)" (Ministry of Agriculture, Forestry and Fisheries of Japan)

The logo of the thinka products has been designed to remind us animals
and it expresses a lovely side of animals. Red is used to show our strong
sense of responsibility for animal medical treatment.
The thinka Canine Heartworm Ag Test Kit CHW will be displayed at the 16th Annual Meeting of Japanese Board of Veterinary Practitioners 2014.
Period: 26 (Fri) - 28 (Sun) September 2014
Venue: Hotel New Otani Tokyo *The exhibition starts on 27 (Sat) at 12:00.
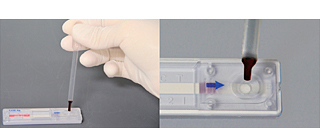
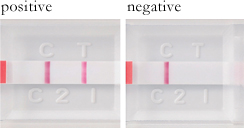
The product allows easy and quick filariasis antigen test during the busy season.
•Operating method, determination method
| Name | thinka Canine Heartworm Ag Test Kit CHW |
|---|---|
| Release date | 27 September 2014 (Sat) |
| Specifications | |
| Target animal | Dogs |
| Sample type | Whole blood, serum, plasma |
| Meas. principle | Immuno Chromatography |
| Reaction time | 5-10 minutes |
| Sample volume | Approx. 80µL |
| Storage | 2-30C |
| Shelf life | 24 months after manufacturing |
| Tests per pack | 10 cartridges, 10 single-use pipettes |
| Approval No. | No. 25-57 |
| Classification | Veterinary medical product, in vitro diagnostic product |
This product will be sold through ARKRAY Marketing, Inc. (ARKRAY, Inc.'s distributor in Japan).
![]()